-

-
Courses
Find courses by:
Collections
Cross-Disciplinary Topic Lists
- About
- Donate
- Featured Sites
This is an archived course. A more recent version may be available at ocw.mit.edu.
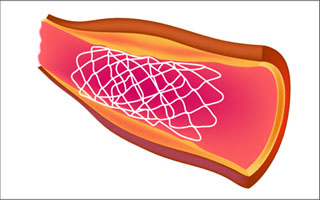
Medical stents are used to keep hollow structures in the body (such as veins and arteries) open. They can be designed to release a drug over a period of time to aid in their effectiveness. (Illustration courtesy of MIT OpenCourseWare.)
Prof. Charles Cooney
Dr. Robert Rubin
Stan Finkelstein, MD
Prof. Tom Allen
Prof. Anthony Sinskey
10.547J / 7.547J / 15.136J / HST.920J
Fall 2005
Graduate
This course serves as a description and critical assessment of the major issues and stages of developing a pharmaceutical or biopharmaceutical. Topics covered include drug discovery, preclinical development, clinical investigation, manufacturing and regulatory issues considered for small and large molecules, and economic and financial considerations of the drug development process. A multidisciplinary perspective is provided by the faculty, who represent clinical, life, and management sciences. Various industry guests also participate.
Charles Cooney, Robert Rubin, Stan Finkelstein MD, Tom Allen, and Anthony Sinskey. 10.547J Principles and Practice of Drug Development, Fall 2005. (Massachusetts Institute of Technology: MIT OpenCourseWare), https://ocw.mit.edu (Accessed). License: Creative Commons BY-NC-SA
For more information about using these materials and the Creative Commons license, see our Terms of Use.
